Research background
Supercapacitors are a new type of energy storage devices developed in the 1970s and 1980s between traditional capacitors and batteries. The current research and development of chemical energy storage systems mainly include: alkali manganese battery, zinc silver battery, lead-acid battery, nickel-cadmium battery, nickel-metal hydride battery, lithium ion battery and so on. Lead-acid battery box lithium-ion batteries have been widely used, they have the following shortcomings: lead-acid batteries have lead pollution problems, as well as large volume and weight, memory effect and other shortcomings seriously restrict its further development; The disadvantages of lithium-ion batteries are relatively high cost, poor safety, and harsh assembly conditions. Under this situation, driven by the application needs of advancing with The Times and the rapid development of science and technology, supercapacitors (electrochemical capacitors) comply with the development requirements of The Times and successfully enter people's vision, becoming another new research hotspot in recent years.
As an efficient, environmentally friendly and relatively new type of energy storage component, supercapacitors have the advantages of high power density, long charge and discharge cycle life, short charge time and long storage life, so they have broad application space and development prospects in consumer electronics, rail transit, aerospace, military, national defense and other fields, and have attracted great attention worldwide. It has become one of the important frontier technologies in the field of energy.
Canonical study
The supercapacitor consists of three parts: electrode material, electrolyte and diaphragm. Electrode materials are the core components of supercapacitors (electrode materials mainly include carbon materials, metal oxides and organic polymers, among which metal oxides and organic polymers have poor cycling stability), which play a key role in the performance of supercapacitors. The research and development of electrode materials with excellent electrochemical performance is the most core topic in the research of supercapacitors.
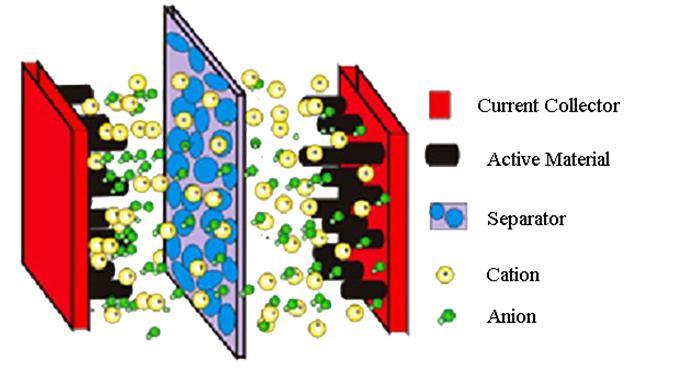
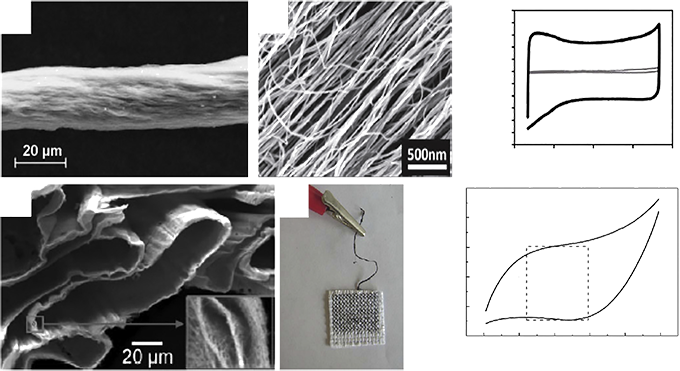
Carbon materials meet the requirements of high electrochemical double layer capacitance due to their high specific surface area (1000 ~ 2000 m2.g-1), high conductivity, electrochemical stability and open pore structure. Carbon materials are widely concerned by academia and industry because of their good charge-discharge stability, and are also the only industrial supercapacitor electrode materials. The electrode materials of supercapacitors are carbon materials (such as activated carbon, activated carbon fiber, carbon nanotubes, carbon aerogel and graphene, etc.). At present, the research of supercapacitor materials mainly focuses on carbon nanotubes, graphene and flexible substrate materials.
1. Carbon nanotubes: Due to low resistance, high specific surface area, unique pore size distribution, and good mechanical and thermal stability, carbon nanotubes have attracted great interest as electrode materials for supercapacitors since the 1990s. Carbon nanotubes can be divided into single-walled carbon nanotubes which are seamlessly coiled by a single sheet of graphene and multi-arm carbon nanotubes which are coaxial nested by multiple single-walled carbon nanotubes.
2. Graphene composites: Graphene has a variety of excellent properties, as well as it can provide a larger theoretical specific surface area for electrochemical reactions, and many of its characteristics are unmatched by other carbon materials, so graphene as a support material with metal oxide or conductive polymer composite used as a supercapacitor electrode material, for electrochemical performance will be greatly improved. The application prospect is also broader.
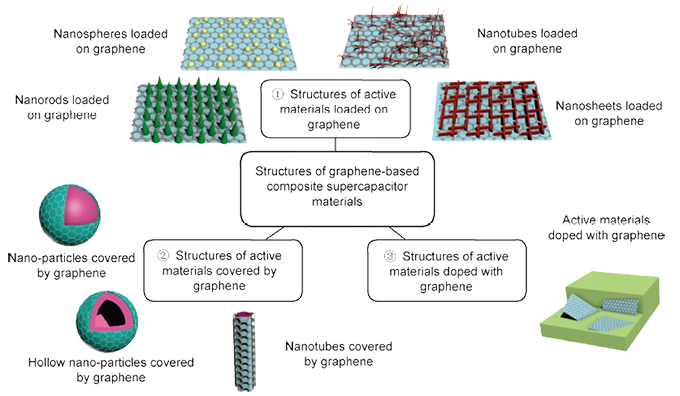
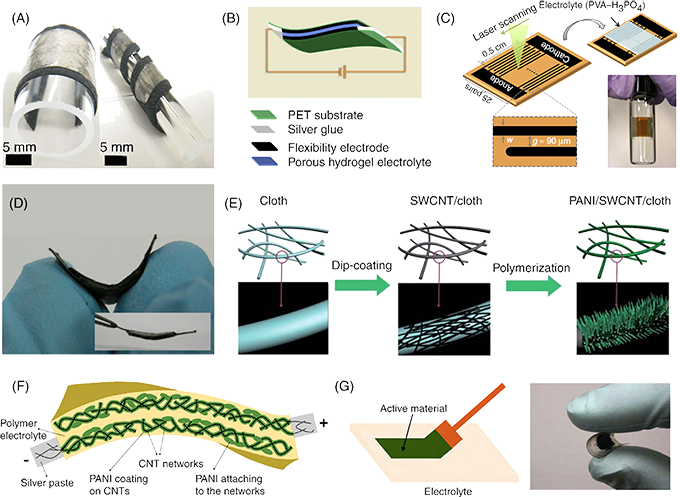
3. Flexible substrate composite material: Flexible supercapacitors mainly refer to the deposition of electrode active substances on the flexible substrate to form electrodes, and then assembled into supercapacitors. Flexible supercapacitors are simpler and do not require a separate collector, because the flexible electrode is electrically conductive and can be used as both a collector and an electrode material. Many materials are currently used as flexible substrates for supercapacitors, such as polyethylene terephthalate (PET), carbon nanotubes (CNT), carbon nanofibers (CNF), graphene films, fabric fibers, and so on.
Equipment recommendation
The EC701C supercapacitor analysis system is used to test the material properties of supercapacitors, mainly to measure current or potential changes. The use of AES-4TH to control the environment of the test system can avoid the interference of external environment (temperature, humidity, etc.) to the system, and improve the repeatability of the measurement.
1. Measuring equipment
EC701 Supercapacitor analysis system: provides current-potential analysis, potential-time analysis, impedance analysis, etc. The equipment has stable performance and high measurement accuracy. For the test of single electrode, the three-electrode method is used. For the assembled supercapacitor system, a two-electrode system is used for electrochemical measurement, the specific method is as follows: for the asymmetric supercapacitor system, the auxiliary electrode and the reference electrode are combined and connected with the negative electrode of the assembled capacitor, while the working electrode is connected with the positive electrode of the assembled capacitor; For a symmetric supercapacitor system, the auxiliary electrode and the reference electrode are combined and connected together with one electrode of the assembled capacitor, while the working electrode is connected with the other electrode.
2. Environmental control equipment
AES-4TH Temperature and Humidity Test Platform: For material or device research to build a temperature, humidity, pressure, atmosphere controlled test environment, the system equipped with precision measurement module, can be used to measure resistance, voltage, current signals, to comprehensively characterize the electrical signal changes of materials, through the whole circuit, line, cavity shielding and anti-interference processing, to achieve very low signal stability test. The equipment is used to provide a stable working environment for the test environment and ensure the repeatability of the experiment.
3. Accessories consumables
a. Flexible substrate materials: polyethylene terephthalate (PET), carbon material substrate (carbon nanotubes (CNT), carbon nanofibers (CNF), graphene films), etc.
b. Electrode material: fluid collecting material, working electrode, reference electrode, counterelectrode and electrolytic cell, etc.
Typical result
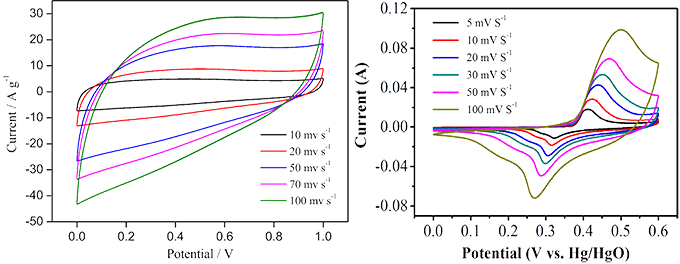
Cyclic voltammetry (CV) : cyclic voltammetry is a basic method to study the electrochemical properties of materials. It is of great significance to study and explore the adsorption/desorption reaction, oxidation/reduction reaction and other behaviors on the electrode surface, as well as the degree of reversibility. Cyclic voltammetry is a method to record the change of current with potential by scanning the electrode potential through different scanning rates and making it scan in the form of triangular wave over time. One or more closed curves are often obtained by this method. The principle of selecting the potential window is that oxidation and reduction reactions can occur alternately on the electrode, so that the performance of the electrode material can be judged according to the shape of the curve. For example, the degree to which the electrode reaction is reversible, the form in which the reaction takes place at the electrode surface (e.g., double layers, pseudocapacitors, Or both), what is the REDOX state of the pseudocapacitor material occurring on the electrode surface (judged by the number of REDOX peaks), what factors control the electrode reaction process (such as diffusion control, charge transfer control, adsorption control or a combination of factors), and the stability of the electrode material under multiple cycle tests.
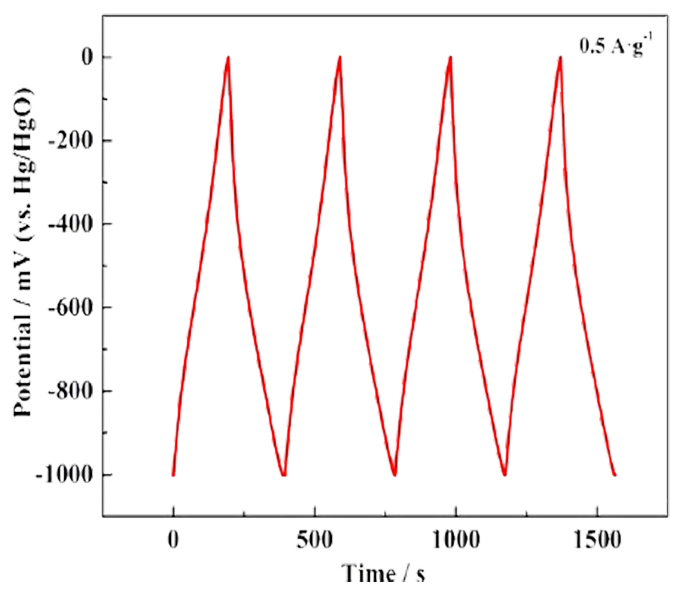
Galvanostatic charge/discharge (GCD) : The specific capacity, rate performance and cycle stability of the electrode material of the supercapacitor are the main indicators to measure its electrochemical performance, which can be achieved by constant current charge/discharge test. The basic principle of constant current charge and discharge is that under the condition of a certain current density, the potential curve of the working electrode for the reference electrode in the process of charge and discharge is studied over time. If the capacitance is constant, then the potential changes linearly over time, that is to say, the constant current charge and discharge curve of the ideal capacitor is a straight line. The charge-discharge curve has a symmetrical triangular distribution, and there is a good linear relationship between voltage and time, which is a typical double-layer capacitor feature and has good reversibility.
Electrochemical impedance spectroscopy (EIS) : EIS is an important means to study the dynamics of electrode processes and surface phenomena. The performance parameters of the electrode were obtained according to the variation of AC impedance. The EIS graph is a curve of the frequency response of the electrode and the electrolytic liquid system, which reflects the resistance and capacitance characteristics of the electrode and the electrolytic liquid system to a certain extent. At the same time, during the test process, the damage to the electrode surface state is not serious, which is one of the commonly used electrochemical analysis tests. Electrochemical impedance spectroscopy is a kind of electrochemical measurement method to measure the corresponding current signal with the small amplitude sine wave voltage of frequency change as the disturbance signal. By analyzing and fitting the electrochemical impedance spectrum results, the equivalent circuit of the electrochemical process can be inferred, and the parameters of the relevant components in the equivalent circuit, such as the equivalent series capacitance and charge transfer resistance, can be calculated, and then the impedance characteristics and kinetic properties of the electrode structure in the electrochemical process can be analyzed. The open circuit voltage of the working electrode is usually tested before the EIS test, and the last value of the open circuit voltage is used as the initial value of the AC impedance test.
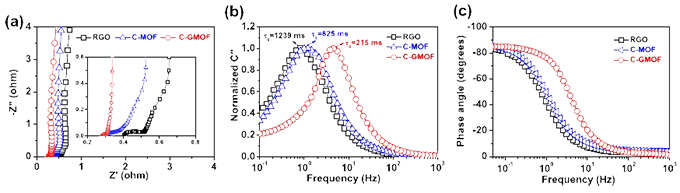
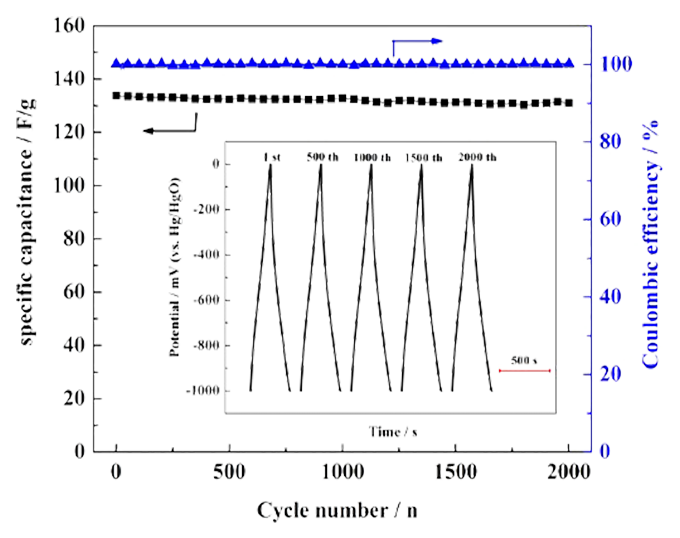
Cycle stability: Cycle life is an important index to measure the performance of supercapacitors. The cycle life of supercapacitors is mainly measured by repeatedly charging and discharging supercapacitors for a long time, and the performance parameters such as capacity, internal resistance and self-discharge are analyzed with the change of charging and discharging times. The biggest advantage of supercapacitors compared with batteries is the long cycle life, which can reach 500,000 to 1,000,000 times. Therefore, for supercapacitors, the measurement of cycle performance is a very important parameter for electrode materials. Generally, the stability of the active material can be judged by repeated cyclic voltammetry tests at a fixed scanning speed, and the difference of specific capacity before and after the cycle can be calculated. It can also be reflected by continuous constant current charge and discharge test at a certain current density. By processing the cycle data, the relationship curve between the capacity retention rate and the number of cycles is obtained, and then the stability of the active material is intuitively displayed. In short, with the increase of the number of tests, the slower the decline of the physical quantity investigated, the better its cyclic stability. By analyzing the morphology, structure and chemical composition of the electrode before and after the test, we can know the main factors leading to the degradation of the electrode during the cycle.
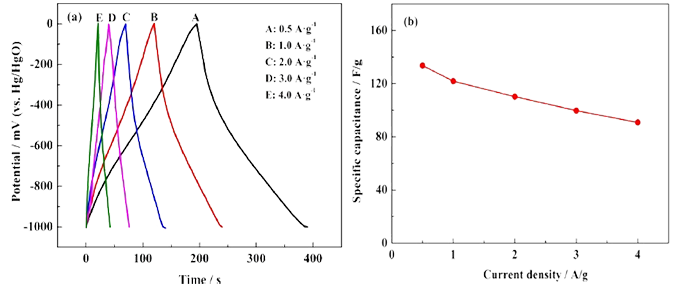
Magnification performance: Magnification performance is the reaction of specific capacitance, retention rate and recovery ability of the electrode material under different current densities. Rate performance is also known as high current charge and discharge performance, mainly through the cyclic voltammetry curve and constant current charge and discharge curve and the obtained capacity (specific capacity) to judge. The magnification performance is mainly related to the reversibility of the reaction on the surface of the electrode material, and the reversibility is related to the stability of the electrode material. The reversibility of charge and discharge of supercapacitors reflects the ability to store charge to a certain extent, so reversibility and magnification are important performance indicators. With the increase of current density, the shape and symmetry of the charge and discharge curve did not change, and no obvious voltage drop was observed on the discharge curve, which indicated that the internal resistance of the electrode material was small, indicating that the electrolyte ions diffused rapidly on the surface of the electrode material, which was related to the porous structure inside the material. In general, if the size of the current response in the cyclic voltammetry curve can increase with the increase of the scanning rate, and the shape of the curve does not undergo large deformation (that is, the polarization is small at high scanning rate), it indicates that the magnification performance of the electrode or device is good. On the other hand, it is to judge how fast the capacity (specific capacity) decreases with the increase of the scanning rate or charge and discharge current, and if the retention rate is relatively high, it indicates that the electrode or device has good rate performance.
SEM and TEM: Describe the surface morphology and internal structure and morphology of the material. SEM scanning electron microscopy can directly perform microscopic imaging on the surface of the sample, so the size, morphology and external structure of the sample particles can be observed. TEM Transmission electron microscopy (TEM) can observe the size, morphology and internal structure of the sample particles.
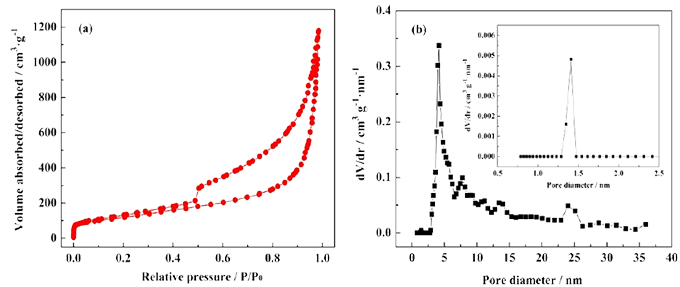
N2 adsorption-desorption: Determine the specific surface area, pore volume and pore size distribution of the material.
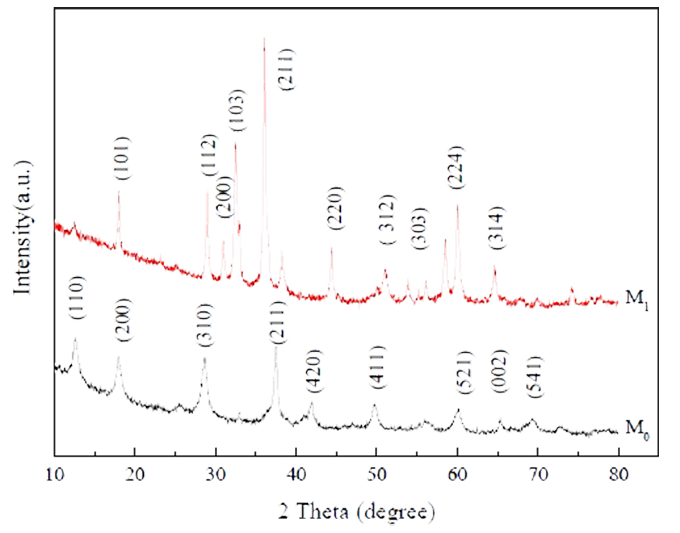
XRD: indicates the material structure and surface adsorption state. XRD analysis is a basic analysis method of inorganic solid materials, it is by analyzing the crystal pattern and comparing with the standard spectrum, so as to judge the substance's crystal pattern analysis test method. The crystal structure and cell parameters of different substances are different, so the diffraction pattern is also different, so the material composition, crystal structure and crystal strength can be analyzed according to the diffraction pattern obtained from different diffraction angles. XRD is one of the most commonly used basic test methods in physics, materials science, chemistry and other research materials.
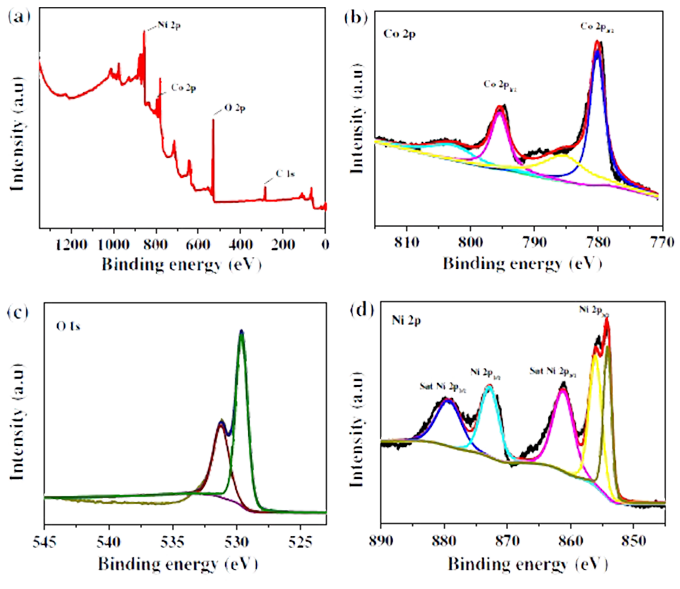
XPS: One of the test methods widely studied and used in materials science, physics, chemistry and other departments in recent years. The test method is superior to other test methods for its comprehensive features such as wide application, non-destructive measurement and semi-quantitative research. It has a very prominent application in the research of chemical composition and element state on the surface of electrode materials.
reference
C.N.R. Rao, A.K. Sood, R. Voggu, K.S. Subrahmanyam, Some Novel Attributes of Graphene, J. Phys. Chem. Lett., 2010, 1, 572-580.
C.N.R. Rao, A.K. Sood, K.S. Subrahmanyam, A. Govindaraj, Graphene: the New Two-Dimensional Nanomaterial, Ange. Chem. Edit., 2009, 48, 7752-777.
P. Li, E. Shi, Y.B. Yang, Y.Y. Shang, Q.Y. Peng, Carbon Nanotube-Polypyrrole Core-Shell Sponge and its Application as Highly Compressible Supercapacitor Electrode, Nano Res., 2014, 7, 209-218.
D.P. Dubal, G.S. Gund, C.D. Lokhande, R. Holze, Decoration of Spongelike Ni(OH)2 Nanoparticles onto MWCNTs Using an Easily Manipulated Chemical Protocol for Supercapacitors, ACS Appl. Mater. & Interfaces, 2013, 5, 2446-2454.
L. Wang, D.L. Wang, Preparation and Electrochemical Characterization of MnOOH Nanowire-Graphene Oxide, Electrochim. Acta., 2011, 56, 5010-5015.